- Your cart is empty
- Continue Shopping
Product
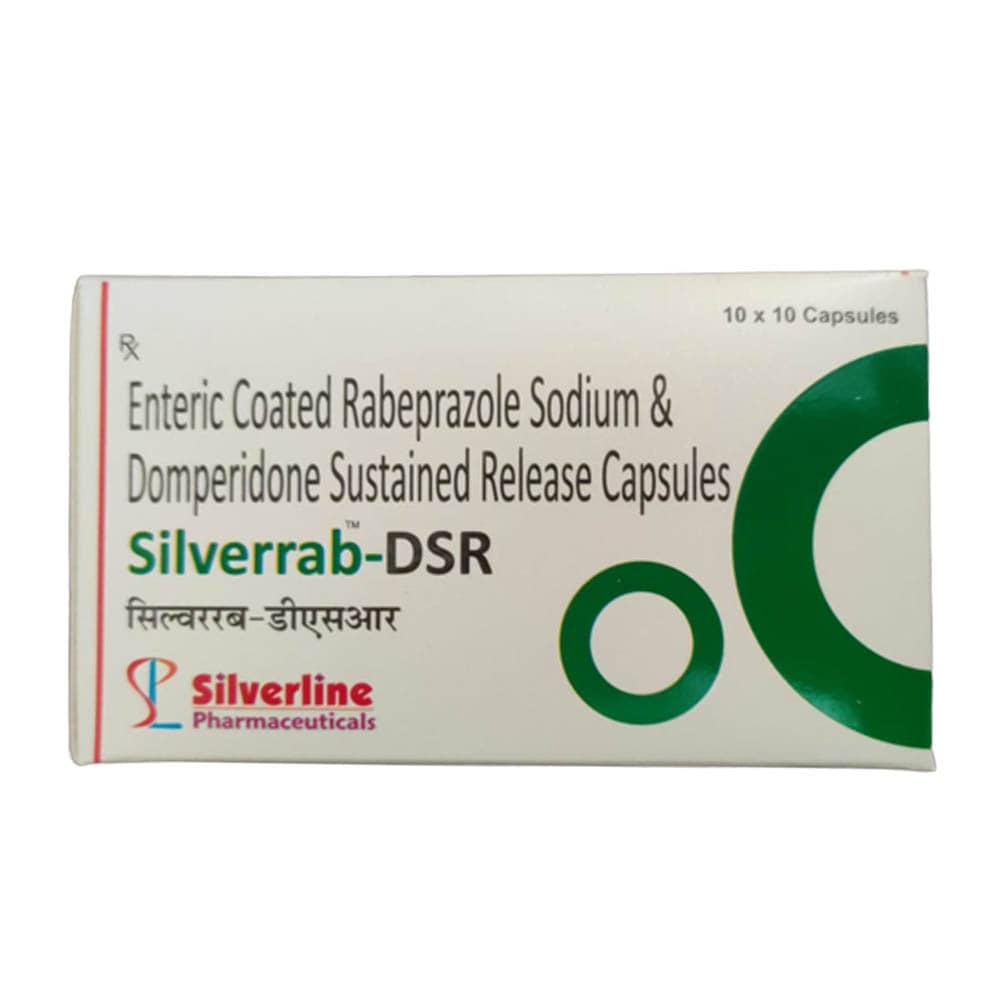
Enteric Coated Rabeprazole Sodium and Sustained Release Domperidone Capsules
| Brand | siilverline pharma |
| Manufacturer | Siilverline pharma |
| Treatment | Treat certain stomach and esophagus problems (such as acid reflux, ulcers) |
| Form Of Medicine | Capsule |
| Packaging type | Box |
| Packaging Size | 10 x 10 Capsules |
| Medicine Type | Allopathic |
| Minimum Order Quantity | 50 Box |
Absorption
T max is 2 to 5 h. Oral bioavailability is approximately 52%.
Distribution
Protein binding is 96.3%.
Metabolism
Extensively metabolized in liver by CYP3A to sulphone metabolite and CYP2C19 to desmethyl rabeprazole. Thioether and sulphone metabolites are formed by reduction of rabeprazole. These metabolites do not have significant antisecretory activity. CYP2C19 exhibits genetic polymorphism caused by deficiency in some subpopulations (white patients, 3% to 5%; Asian patients, 17% to 20%).
Elimination
Plasma half-life is 1 to 2 h. Eliminated in urine (90% as thioether carboxylic acid, glucuronide, and mercapturic acid); remainder recovered in feces. No unchanged drug recovered.
INDICATION : The management of dyspepsia, heartburn, epigastric pain, nausea, and vomiting.
DOSAGE: As directed by the physician.
SIDE EFFECTS: Irritability, Dry mouth, Headache, Loss of interest in sex, Breast pain or tenderness, Abnormal heart rhythm, Drowsiness, Abnormal milk discharge from breast, Diarrhoea.